Set procedures at the start to save costs and achieve efficient production. The aim is to set clear criteria and guidelines for :
- Standardisation of fonts
- Proper forming of braille characters
- Standardisation of positioning
- Testing and controlling
Standard for Braille fonts
Braille Cell Dimensions
 It is recommended that the Marburg Medium font specification is used for pharmaceutical packaging and labels. Marburg Medium details the size, spacing and positioning of the braille dots. PharmaBraille fonts follow this specification.
It is recommended that the Marburg Medium font specification is used for pharmaceutical packaging and labels. Marburg Medium details the size, spacing and positioning of the braille dots. PharmaBraille fonts follow this specification.
Braille Character Codes
Braille codes consist of letters, numbers, punctuation, symbols and special characters. Some parts of character sets are common between countries whereas other parts differ, e.g. Latin versus other alphabets and accented letters.
In the artwork creation process, the braille code set to be used should be verified as appropriate for the country in which the medicinal product is to be supplied. The Marketing Authorisation Holder (MAH) and packaging supplier must check all Braille artwork for current accuracy and relevance. The EBU PharmaBraille Codes & Guidance area of our website contains information on European braille codes. In addition the International Blindness Agencies Directory can help to identify Braille organisations for each country.
If multi-market, multilingual packs are being produced with braille text, the correct character sets should be identified and included in the artwork.
Capitalization should be avoided and trademark symbols omitted as these take up unnecessary space.

Technical standards of the Braille form
Braille legibility
It is important that the braille dots are produced strongly enough for the blind person to identify the product. The braille cell dot height limits, agreed between the MAH and packaging supplier, must be achieved to ensure braille text legibility.
Braille cell dot height
The CEN standard refers to a dot height target of 0,20mm
Standard Layout Specifications
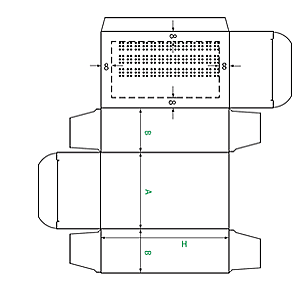 It is necessary for the MAH and the packaging supplier to agree the braille text to be applied, and its placement on the packaging material.
It is necessary for the MAH and the packaging supplier to agree the braille text to be applied, and its placement on the packaging material.
Braille can be placed on any main side of a folding carton. The ideal position from a production point of view is to have the embossing on the side next to the glue flap. However, production set-ups vary, so the carton manufacturer should be consulted before braille artwork is completed.
The braille may be positioned over printed text or graphic elements, but should avoid areas containing creases, barcodes, labels, perforations etc. It is suggested that the distance from the edge of the dot to cutting lines, creasing lines, perforations etc. should be no less than 8mm.
Control of Artwork Files and Print Approvals
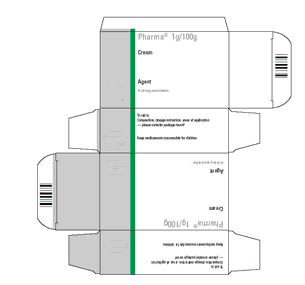 It is important that the print artwork and the Braille content are clearly communicated on the packaging artwork. This needs to be agreed between the MAH and the packaging supplier. Once approved the artwork should not be amended unless agreed by all parties. Formats for submission of artwork containing braille can vary for each regulatory authority.
It is important that the print artwork and the Braille content are clearly communicated on the packaging artwork. This needs to be agreed between the MAH and the packaging supplier. Once approved the artwork should not be amended unless agreed by all parties. Formats for submission of artwork containing braille can vary for each regulatory authority.
There might be several layers of artwork to define the packaging (e.g. text and graphics, braille, die-line, varnish, anti-counterfeiting measures). For clarity and verification, the colour used to represent the braille text should not be used in any other place in the artwork.
The approval of the proof should confirm that the braille characters are correct. It is important that the local braille authority is consulted to ensure that the correct local braille characters have been used. The EBU PharmaBraille Codes & Guidance area contains information on European braille. The International Blindness Agencies Directory can help to identify braille organisations for each country.
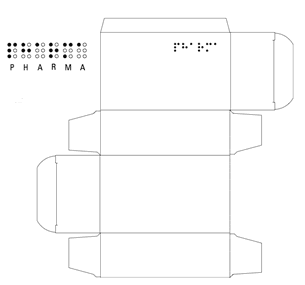 A translation of the braille text may be reproduced outside the die-line for quality assurance purposes.
A translation of the braille text may be reproduced outside the die-line for quality assurance purposes.
It is recommended that digital approved artwork for Braille packaging is established with separate layers or pages – in particular for separating the braille text from the printed graphics and text, as braille may be positioned on top of these elements.
For printed paper proofs it is recommended that the approved artwork proof should be set up as follows:
- on the first page all texts, graphic elements and die-line except for the braille text should be presented
- on the second page only the braille text and the die-line should be presented
Sources:
- European Standard Developments – CEN. Standard EN 15823
- European Blind Union
- International Association of Diecutting and Diemaking
- European Braille Guidance
- “Braille Cell Dimensions”, Tiresias – Scientific and Technological Reports
- “Braille on folding Cartons”, European Carton Makers Association, Sept 2005
- EN 15823:2010 “Packaging. Braille on packaging for medicinal products”
- University of Birmingham, “Braille dot height research: Investigation of Braille Dot Elevation on Pharmaceutical Products”
Note:While every care has been taken to check the accuracy of the braille symbols we cannot guarantee the accuracy of this information. Please confirm braille character sets with the relevant local braille authority. The International Blindness Agencies Directory can help to identify Braille organisations for each country.
Note:The information above is for guidance only. Please confirm technical and artwork standards with the relevant local regulatory body/ies.